Professor James Economy's Group
|
|
[Activated Carbon Fibers] [Ion Exchange Fibers] [Chelating Fibers] [Membranes] [Porous Inorganic Fibers]
Polymeric
Ion-Exchange Fibers This
work explores the design of new ion-exchange materials in
the form of fibers that yield a number of important
advantages over conventional ion-exchange beads. In this
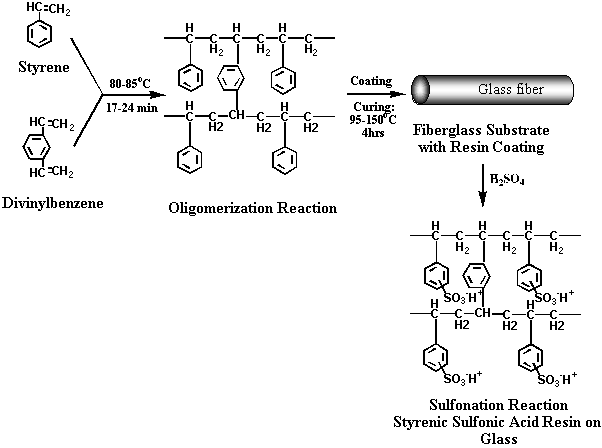
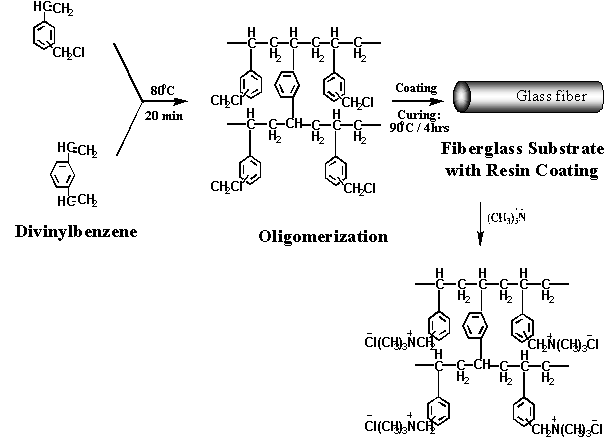
approach, ion-exchange fibers are prepared by (1) coating
low-cost glass fiber substrates with an appropriate
oligomer, (2) cross-linking, and (3) functionalizing the
coating to produce either anionic or cationic capability
(see Figure 1 and Figure 2). As a result
of the thin coatings, the use of solvents prior to both
functionalization and preswelling of the finished product
prior to end-use was eliminated, representing a
significant simplification of current synthesis methods.
Kinetic experiments showed that the contact efficiencies
of these systems were greatly improved over the
traditional beads because of the higher surface-to-volume
ratio and shorter diffusion path lengths. This
improvement translated into an order of magnitude
increase in both ion-exchange and regeneration rates.
Another advantage is the excellent resistance of the
fibers to osmotic shock even after multiple regenerations
(see Figure 3).
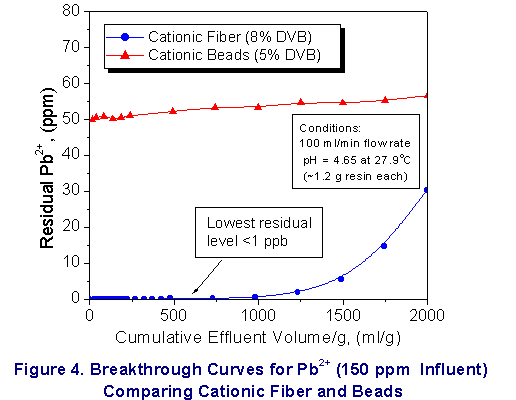
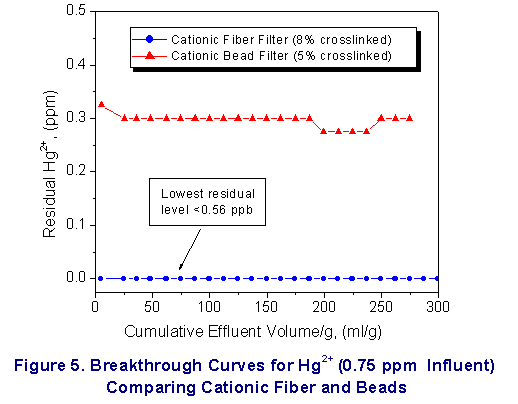
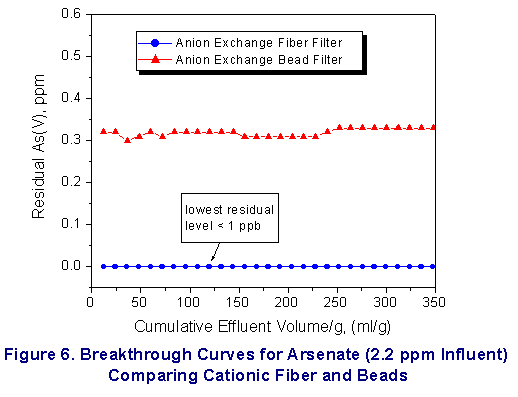
Finally, these systems were shown to remove heavy metal
contaminants ( Hg2+, Pb 2+ and arsenate
ion )effectively to well below part per billion
concentrations (see Figure
4-6). Key Features as Compared
to Beads: • Simplified synthesis
(1/2 the steps) Figure
1. Synthesis of cationic fibers on glass substrate Figure 2. Synthesis of
anionic fibers on glass substrate Ion Exchange
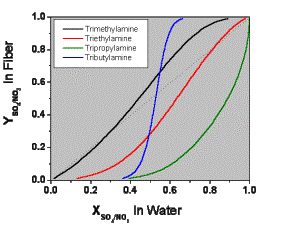
Fiber Selectivity
References: 1.
Dominguez, L., J. Economy, K. Benak, C. Mangun, “Anion
exchange fibers for arsenate removal derived from a
vinylbenzyl chloride precursor,” Polym. Adv.
Technol. 14, 632-637, 2003. 2.
Dominguez, L., Z. Yue, J. Economy, C. Mangun, “Design
of polyvinyl alcohol mercaptyl fibers for arsenite
chelation,” Reactive and Functional Polymers
53(2-3), 205-215 2002. 3.
Economy, J., L. Dominguez, C. Mangun, “Polymeric ion
exchange fibers,” Industrial & Eng. Chemistry
Research 41(25), 6436-6442, 2002. 4.
Economy, J., C. Mangun, “Novel fibrous systems for
contaminant removal,” In Sampling and Sample
Preparation for Field and Laboratory, Ed. J. Pawliszyn,
Elsevier Science, 2002. 5.
Benak, K., L. Dominguez, J. Economy, C. Mangun, “Sulfonation
of pyropolymeric fibers derived from phenol-formaldehyde
resins,” Carbon 40(13), 2323-2332, 2002. 6.
J. Economy. “Now that's an interesting way to make a
fiber!” CHEMTECH 10(4), 240-7. 1980 |
||||||||||||||||||||||||||||||||||||||||||||||||||||||