Professor James Economy's Group
|
|
[Bactericides Materials] [HDD Lubricants] [New materials for Fuel Cell] [Gas Hydrates] Highly
Effective Bactericides Based on Silver Coated Fiber Glass Activated
carbon has been widely used in water purification due to
its high adsorption rates and capabilities. The inherent
drawback of carbon materials, however, is that they have
excellent biocompatibility with bacteria. Bacteria may
breed on carbon during the purification process, thus
making carbon materials themselves pollutants. To get
around this problem, some researchers have explored
depositing colloidal-sized silver, a wide-spectrum
antibiotic, onto carbon substrate as bactericides1-5.
Our experiments showed there are a number of problems
with this approach. First of all, the silver coated
activated carbon has limited antibacterial activity.
Secondly, high temperatures required for carbonizing and
activating activated carbon fibers cause silver particles
to coalesce and form large particles that have inferior
antibacterial activities. Thirdly, severe attrition of
silver particles not only cause antibacterial activities
to deteriorate dramatically, but may potentially cause
“Argyria” as well. In this
study, we developed a new kind of antibacterial fibers by
chemically coating fiber substrates of glass with
colloidal sized silver particles. The antibacterial
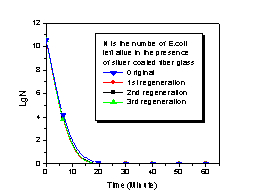
activities were evaluated against E.coli and
compared with their regenerability has been investigated
as well. Antibacterial
fibers based on activated carbon fibers were prepared
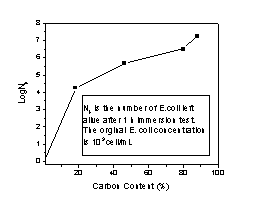
with different carbon contents. As shown in Figure 1,
while silver contents remained at 3%(wt), the higher the
carbon content, the higher the number of E.coli
left live, the less efficient these fibers were as
bactericides. Silver coated activated carbon fiber with
18% carbon content killed up to 4 orders of E.coli
in the contaminated water, while with 90% carbon content,
only minimal bioactivity was observed. It appeared the
uncovered carbon surface still has excellent affinity to
bacteria. To eliminate bacteria breeding, fiberglass was
chosen over activated carbon fibers as fiber substrate,
and quantitative removal of E. coli was achieved. A related
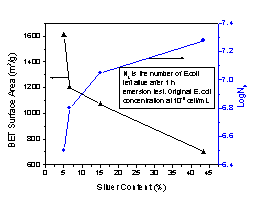
study on the effect of silver content of activated carbon
fibers on their antibacterial activities revealed
activated carbon has very limited tunability as fiber
substrate. ACF-15 was coated with various amount of
silver, as can be seen from Figure 2, BET surface area of
these antibacterial fibers decreased dramatically as
silver content increased, and larger silver particles (1
~ 10um) were more likely to form on the activated carbon
surfaces. As a result, biological activities against E.
coli decreased, but only slightly. This suggested
that antibacterial activities of silver coated activated
carbon fibers were not sensitive to fiber properties,
such as surface area and silver content, which made it
difficult to tailor the antibacterial activities. As
comparison, antibacterial fibers made from fiberglass
substrate were easily modified by changing silver
content, fiber diameter and surface morphology to achieve
quantitative removal of bacteria in minutes.
References 1.
Oya, A. and Yoshida, S. (1993). Antibacterial activated
fiber derived from phenolic resin containing sliver
nitrate. Carbon 31, 71-73 2.
Oya, A., Wakahara, T. and Yoshida, S. (1993). Preparation
of pitch-based antibacterial activated carbon fiber. Carbon
31, 1243-1247. 3.
Oya, A., Kimura, M., Sugo, T., Kataki, A., Abe, Y.,
Iizuka, T., Makiyama, N., Linares-Solano, A., and
Salinas-Martinez de Lecea (1994). Antibacterial activated
fiber derived from methyl methacrylate-grafted phenolic
resin fibers. Carbon 31, 107-110. 4.
Li, Ch.Y., Wan, Y.Z., Wang, Y.L., Jiang, X.Q., and Han,
L.M. (I998) Preparation and Characterization of
antibacterial pitch-based activated carbon fiber
supporting silver. Carbon 36, 61-65. 5.
Wang, Y.L., Wan, Y.Z., Dong, X.H., Cheng, G.X., Tao,
H.M., and Wen, T.Y. (I998) Preparation and
Characterization of antibacterial viscose-based activated
carbon fiber supporting silver. Carbon 36,
1567-1571. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||