Professor James Economy's Group
|
|
[Carbon/BN Composite] [AlB2 Composite] [Polymer Composites]
Aluminum
/ Aluminum Diboride Metal Matrix Composites In-situ
aluminum matrix composites based on the
aluminum-copper-boron ternary system have been developed
for the first time. Aluminum diboride, one of many stable
crystalline phases naturally present in the alloy system,
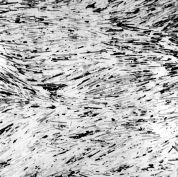
is exploited as a reinforcement. AlB2 exists
in the aluminum matrix as single crystal high aspect
ratio flakes. Proper heat treatment allows for the
preparation of an aluminum composite containing only this
phase. Reinforcement volume fraction can be varied from 0
to 20% by control of alloy composition and by the use of
a modified squeeze casting apparatus. At 20v/o loading of
AlB2 an 80% increase in strength over the
matrix alloy is observed. Wear and creep resistance of
these composites is also found to be excellent. Current
research is focusing on the following areas: Redtermination
of the Al-B phase diagram. Considerable
disagreement exists in the literature concering the Al(L)
+ AlB2 -> Al(L) + AlB12
peritectic transition temperature. Experiments to
determine this temperature are underway. Preparation
of AlB2 MMC's for comodity
materials. Using
comodity boron sources we project that 20 % AlB2
MMC's can be prepared for less than $5/Kg. Preparation
of macroscopic oriented boride structure in aluminum. Macroscopic
structures of oriented borides have been created in
aluminum. It is believed that these structures could lead
to composites with unique properties. Selected
Micrographs Related
Publications The
following is a list of related publications which you may
find of interest. 1.
Deppisch, G. Liu, J.K. Shang, and J. Economy; Fabrication
of AlB2 Flake Reinforced Al-Alloy Composites 2.
C. Deppisch, G. Liu, J.K. Shang, and J. Economy; Effect
of Cooling Rate on the Crystallization of AlB2 Flakes in
Al Composites 3.
L. Wohrer, A. Wosilait, and J. Economy; SAMPE 18,
340-68 (1973). |
||||||||||||||||||||||||||||||||||||||||||||||||||||||